by Clarence Oxford
Los Angeles, CA (SPX) May 01, 2026
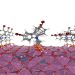
Chemists at the University of Warwick have identified a series of previously unknown materials formed during the heating of molecular precursors, including a new structural form of bismuth vanadate, a well-studied clean-energy compound with potential roles in solar fuel generation and next-generation batteries.
The findings, published April 30 in Nature Communications, show that the intermediate stages between a molecular precursor and its final heated product can themselves yield materials with distinct and useful properties – phases that conventional synthesis methods would not produce.
The team started with specially designed single-source precursors: molecules pre-loaded with all the chemical elements needed to form a target material. By carefully controlling the heating process and tracking transformations in real time, researchers captured several new material phases that would otherwise pass unnoticed.
Among those phases is a previously unknown, kinetically stabilised form of bismuth vanadate designated beta-BiVO4. Standard BiVO4 is valued in clean energy research because its band gap – the energy threshold required to absorb sunlight and drive chemical reactions – is well-matched to solar radiation, enabling the material to split water and generate hydrogen fuel. The newly identified beta variant carries a different atomic arrangement and a significantly larger band gap, meaning it interacts with light differently from known forms of the compound. The researchers say this could offer new degrees of control over performance in solar fuel generation, catalysis, and electronics applications.
A separate intermediate material uncovered during the study showed a capacity to store large amounts of lithium, raising the possibility of applications in next-generation battery technologies.
Dr Sebastian Pike, Department of Chemistry, University of Warwick, said: “When materials are made by heating, scientists usually focus on the final product, the ‘B’ that results from ‘A.’ But this study shows that there are many fascinating stages in between ‘A’ and ‘B,’ and these hidden steps, could be just as important.”
Dr Dominik Kubicki, School of Chemistry, University of Birmingham, said the results reframe how intermediates are viewed: “These ‘in-between’ materials aren’t just stepping stones – they can have useful properties in their own right. By understanding and controlling how they form, we can start to design better materials for batteries, catalysis, and solar energy.”
The characterisation work combined solid-state NMR spectroscopy, X-ray diffraction, and pair distribution function analysis. The team found that the choice of precursor and the specific breakdown pathway it follows can be used as a deliberate tool to steer which structures form, giving access to phases that are inaccessible through standard high-temperature routes.
Dr Pike noted that only a small number of precursors were examined in the current study, but said the approach points to a wider opportunity in materials discovery: precise temperature control and precursor chemistry could unlock many more hidden but functionally useful phases across a range of material systems.
Research Report: Amorphous intermediates and discovery of a kinetic polymorph of BiVO4 from heating V+Bi+Zn single-source precursors
Related Links
University of Warwick
Powering The World in the 21st Century at Energy-Daily.com